To test the REMEDi4ALL platform’s viability and robustness we support real-world repurposing projects, known as ‘demonstrator projects’, covering infectious diseases, rare cancers, and rare/ultra rare diseases. Learn more about the REMEDi4ALL demonstrator projects.
The VESPA Trial – Drug Repurposing for Pancreatic Cancer
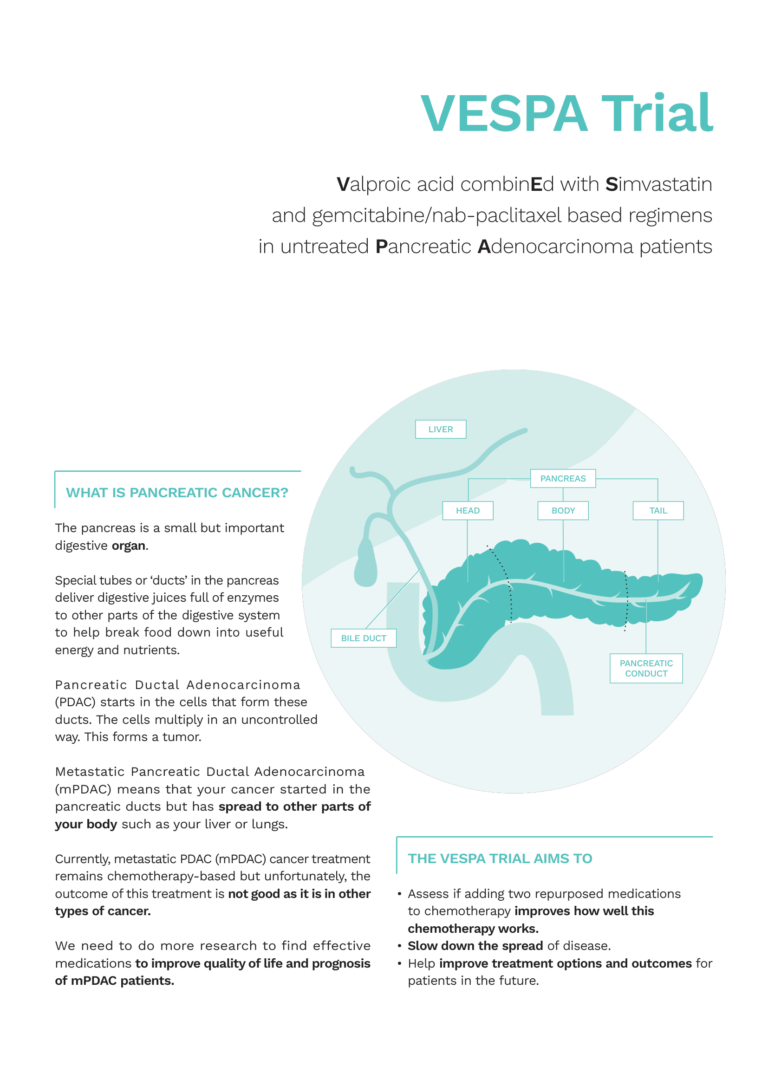
170 patients across Italy and Spain who have been diagnosed with Metastatic pancreatic ductal adenocarcinoma (mPDAC) and are about to start treatment will be invited to take part in VESPA. In the lab, there have been very promising results showing that a combination of Simvastatin (originally used to lower cholesterol) + Valproic Acid (originally used to prevent seizures and in some mental health issues) can improve how well normal chemotherapy treatment works. The three different drugs attack different biological pathways that the cancer relies on to survive.
Both Simvastatin and Valproic Acid have been used by doctors around the world for many years. This means we know they are safe and have minimal side effects. Any side effects that do arise are well documented and understood so can be addressed quickly.
Of the 170 patients, everyone who enrols onto the VESPA trial will receive the chemotherapy treatment that is the standard of care for mPDAC. No one will receive a placebo drug as this would be unethical in this situation and everyone receives the same level of care as if they did not join the trial. Half of the patients will receive the additional repurposed drugs.
Patients will be randomly assigned to receive either the chemotherapy treatment alone (known as the control arm) or the chemotherapy combination therapy (chemotherapy + simvastatin + valproic acid – known at the intervention arm). This study is open label which means that the doctors giving the treatment and the patients receiving the treatment will know which arm they are assigned to.
Key aims of the VESPA trial:
· To determine if using simvastatin and valproic acid in combination with chemotherapy is safe for patients
· To determine if using simvastatin and valproic acid in combination with chemotherapy extends the amount of time before a patient’s cancer progresses (grows or spreads)
The VESPA trial will also look at:
· How the treatment(s) impact patient’s quality of life (QoL)
· How many side effects patients are experiencing and how bad these side effects are
· Looking at specific proteins found in a blood test that could be used to identify pancreatic cancer in the bloodstream
· Looking at pancreatic tumour cells to tell us more about specific proteins and mutations that are present in this type of cancer
Patient engagement in the VESPA Trial – Drug Repurposing for Pancreatic Cancer
Patient Fact Sheet
The patient fact sheet was designed to sit alongside the ‘formal’ Patient Information Sheet (PIS) and Informed Consent Form (ICF), offering a more visual and accessible way of engaging with the trial and what participation might look like for patients.
Multi-Stakeholder Meeting (MSM)
Multi-Stakeholder Meetings (MSMs) bringing together different groups and stakeholders involved in drug development to address some of the challenges and through discussion and networking, aims to provide actions to help overcome some of these challenges.
This collaborative approach allows all stakeholder to understand what their individual and distinct priorities are when thinking about drug development. This is particularly special as often each part of the drug development pipeline occurs in silos where there is limited cross-stakeholder discussion.
Resources
1. Pancreatic Cancer Europe – an European multi-stakeholder platform which aims at bringing together experts from all over Europe including academics, physicians, politicians, patient groups and journalists with a common interest and willingness to improve care for patients with pancreatic cancer. https://pancreaticcancereurope.eu/
2. Pancreatic Cancer UK – “we’ll transform the future for pancreatic cancer by funding research breakthroughs, campaigning for changes to treatment and care and supporting people when they need it the most”. https://www.pancreaticcancer.org.uk/
3. Pancreatic Cancer Action Network – “Our mission is to take bold action to improve the lives of everyone impacted by pancreatic cancer by advancing scientific research, building community, sharing knowledge, and advocating for patients”. https://pancan.org/
4. Nadia Valsecchi Foundation – a non-profit organization with the main objective of supporting scientific research on pancreatic cancer. https://www.fondazionevalsecchi.org/
What is REMEDi4ALL?
Drug repurposing means finding new uses for existing medicines. Instead of developing new drugs, repurposing builds on existing knowledge of a drug’s safety, efficacy, and dosing, which allows for a potentially faster and more cost-effective route to new treatments.
REMEDi4ALL is an EU-funded initiative that aims to develop an accessible, standardised platform to provide the expertise, tools and resources required in all stages of the repurposing journey. REMEDi4ALL also hopes to build a global drug repurposing community and to generate a more favourable policy environment by identifying current barriers and exploring solutions.